Another scientific breakthrough has yet to bridge the divide between science and ethics. Koli Mitra walks the tightrope.
When Star Trek’s Doctor Crusher needed a new body part for a patient – perhaps someone who lost a limb while repairing the star ship’s “warp engine” – she had some fantastic 24th Century technology at her disposal. She could just tweak a “food replicator” by supplying it the molecular and design specifications for a toe, a liver, or an ear.
Now it seems we’ve arrived at the 24th Century well ahead of schedule. Today’s additive manufacturing devices (‘3D printers’) are being used to “print out” everything from the delightful, (violins and chocolate) to the truly disturbing (guns).
How it works is, you send instructions from a computer to a machine, like an inkjet printer, except, instead of making 2-dimensional images with ink sprayed in designated patterns, it takes some substance (polymer sheets, powders, or pulps) and layers it in designated patterns to form a precisely shaped 3-dimensional object.
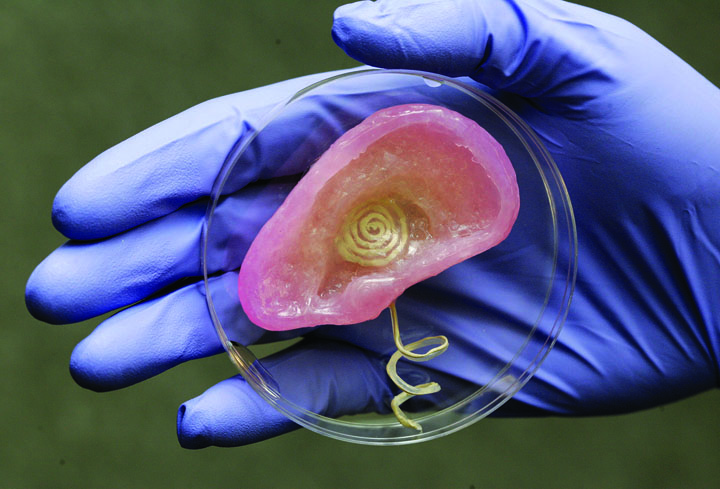
This year, biomedical researchers at Cornell University made a prototype human ear by “printing” a collagenhydrogel mould from 3D image files of a natural ear and seeded it with a cartilage cell culture, which then grew into fully-formed ear cartilage. Right now, only the structure is human; the cells came from animals. But as biochemical processes become easier to automate, like the manufacturing already is, organ printing could become routine. Just as monochromatic document printing soon gave way to multicoloured printing, the earliest 3D printers, which printed only single-substance structures, are giving way to machines that can use multiple materials in numerous combinations, making things that are not only morphologically complex, like musical instruments, but also have to be chemically precise, like food… or both, like body parts.
This has extraordinary implications for regenerative and reconstructive medicine. For centuries, surgeons have tried to replace lost or damaged physical features, by attaching prosthetics, transplanting organs donated by others, and grafting pieces of tissue taken from other areas of patients’ own bodies. But the most painstaking reconstructive surgery can still only create (relatively) crude facsimiles of a limb, digit, or organ that nature seems to produce so effortlessly. If you’re missing an ear, a surgeon can fashion a new one from cartilage found elsewhere in your body, typically the ribs. But ear cartilage has unique properties, giving it a perfect balance of strength and flexibility, ideal for an ear’s functions, which rib cartilage just can’t match.
Some organs – hearts, lungs, pancreases – are too complex to reconstruct from a body’s other tissues. So, transplantation is the only option. But naturally occurring organs aren’t exactly lying around as spare parts. Someone has to die or at least undergo an amputation to give an organ. Moreover, there are serious concerns about organ trafficking and coercion of impoverished and marginalized people. This means organs obtained safely and ethically often come from people who have died from illness or old age and not always in the best condition. Most usable organs tend to be from accident victims – not something we’d like to see increasing. The potential for using animal organs – or xenotransplantation – has been studied, but, so far without success; and it raises profound ethical issues of its own. Also, transplantation (even within our species) comes with a risk that the patient’s immune system – evolved to fight off invasive foreign biological matter – will reject the transplant. So… the prospect of growing new organs from patients’ own cells is getting much interest these days, making “regenerative medicine” the hottest new research field.
Scientists have cultured human cells in laboratories for years, but now, they’re increasingly able to custom-design cells to fit specialised requirements. Human embryonic stem cells can be developed into any cell the body needs. This year, for the first time, patient-specific embryonic stem cells were successfully cloned from adult human cells (which can be implanted into the same adult human without rejection risk, since they’re not genetically “foreign”). Another patient-specific process, now favoured by most researchers, is “induced pluripotent stem cells” (iPS) derived from adult somatic cells by reprogramming them to act like embryonic stem cells. The iPS method eliminates ethically murky issues associated with embryonic stem cells (like cloning and embryo-destruction).
Cells, though, are just building blocks. Once you’ve created some in a petri dish, how do you configure them to form a functioning organ? Among other things, you’ll need a mould – a scaffold – on which the cells could grow to give the organ the right form. In 2011, scientists at the University of Minnesota, led by Dr. Doris Taylor, found a way to chemically dissolve all of the cellular material on a heart without damaging its underlying protein ‘skeleton’. It left behind what Taylor calls a “ghost heart”, an exact matrix of the human heart – down to the finest branches of the most intricate web of capillaries – that can serve as the scaffold for growing a new heart from a patient’s own cells.
Dr. Taylor may not have to strip a real heart down to its skeleton every time, however. She can just keep a complete profile of 3D images of the organ in a computer file and just “print” the scaffolding using appropriate materials. Nobody has successfully printed a heart matrix yet, but the Cornell team did make an ear, and that’s certainly a start.
But is it all good news? 3D printers are already being used to make handguns. What sinister purposes might be served in future when people can manufacture biology at home? Researchers are understandably focused on the therapeutic purposes for which they are developing the technology. But, our moral development as a species hasn’t always kept pace with our dazzling capacity for technical innovation. For now, I guess we’ll just have to play it by ear.


venlo casino bonus aktion
Die Unterschiede zu Einzahlungsboni und Freispielen, für die oftmals eine Einzahlung notwendig ist, liegen hierbei meist im Bonusbetrag oder der Anzahl der
Freispiele. Zusätzlich zum Einzahlungsbonus werden Sie in diesem Fall mit höchstens 100 Freispielen belohnt.
Um Ihnen den Überblick zu erleichtern, und die Suche nach den besten Casino Freispielen zu beschleunigen, präsentieren wir Ihnen eine Liste der Sachen, die es bei einem Freispielbonus zu berücksichtigen gilt.
Die Wahrscheinlichkeit zu gewinnen ist aber mit wenig
Budget eher geringer, da diese hohen Gewinne natürlich weniger häufig
vorkommen. Das Spiel bietet Scatter Symbole welche Freispiele auslösen können, sowie
andere Mystery Symbole und Multiplikatoren. Natürlich gibt es noch viele weitere Slots, die für Freispielangebote genutzt werden können. Es gibt eine große Auswahl an Slots von verschiedensten Spieleanbietern. Die meisten Freispiele haben einen Wert
zwischen 0,10 und 0,20 Euro.
References:
https://online-spielhallen.de/instant-casino-erfahrungen-meine-umfassende-bewertung/
Joo Casino Spielguthaben
Einen Bestätigungs-Code per SMS oder eine sofortige Kontoverifizierung musst
du genau wie bei Wettenlive (Erfahrungen) nicht ausführen. Auszahlungsanfragen werden innerhalb einer Stunde bearbeitet und an den entsprechenden Zahlungsanbieter weitergeleitet.
Würfelspiele wie zum Beispiel Sic Bo sind ebenfalls vertreten. Es
gibt zwar Online Casinos, die eine noch größere Spielauswahl
zu bieten haben, doch mehr als 3000 Slots benötigen ebenfalls gut funktionierende
Kategorien und Filter. Jeden Monat findet eine Lottoziehung statt,
bei der es Echtgeld (10€ bis 500€) und Freispiele (30 bis 100) zu gewinnen gibt.
Ich bin Rolf Brinker und bei Playcasino.com als erfahrener Experte bereits seit 2022 zuständig für
den deutschsprachigen Markt. Wenn dir also ein sicheres und
legales Glücksspiel wichtig ist, dann solltest du dir jetzt gleich mit deiner ersten Einzahlung von 10€ dein 30€ Bonusguthaben und 30 Freispiele für
Lord of the Ocean sichern. Denn hier läuft alles recht
unkompliziert, angefangen von der Registrierung sowie der Verifizierung
des Spielerkontos bis hin zur Einlösung der Boni und den Gewinnauszahlungen,
die meist in Echtzeit erfolgen. Das Wunderino Casino bietet dabei eine ähnliche Spielauswahl im Bereich
der Video Slots an und liegt auch in Bezug auf die Reload Boni und Aktionen gleichauf mit StarGames.
Richtig üppig fällt unserer Meinung nach der 400% Willkommensbonus bei Wunderino
aus. Jedoch bietet dir Tipico Games eine weitaus breitere
Spielauswahl an.
References:
https://online-spielhallen.de/irwin-casino-aktionscode-alles-was-sie-wissen-mussen/
2026 online gambling login
The Darling is in close proximity to the gambling floors of The Star Casino
and many high rollers stay here. Sydney Casino sits comfortably among the best pokies venues in NSW, with
their status protected by a deal which stops Crown Sydney from hosting poker machines
until 2041. The Star sits comfortably among the best gambling
venues in Australia, with it a popular tourist attraction and nightlife destination for locals.
As players climb through the reward tiers, they’ll be entered in to win additional prizes and have more special offers available to them.
The Star Casino comes with a hotel, and both Jupiter’s Hotel and Casino
and the Treasure Casino participate in the program as well.
Room service, valet, and concierge are all ready to ensure that
your stay is in every way first-class. The Star features a duo of individual luxury hotels, The Darling
and Astral Tower & Residences. Because the Star is located geographically
in Sydney’s premium entertainment area, it’s able to provide accommodations that
are uniquely luxurious.
References:
https://blackcoin.co/vip-slots-best-slots-for-real-money-play-with-bonuses-2023/
BTG slots UK
These chicken balls are bathed in a delectable
sweet and sour sauce, but they’re still deliciously crispy.
You can always count on chicken if you’re looking for a leaner version of
fatty, greasy beef patties. Beer can chicken is a unique dish where the bird is
placed over a can of beer and cooked over indirect
heat. They’re perfectly crunchy on the outside and
covered with the most drool-worthy buffalo sauce.
Marry Me Chicken is a creamy pasta dish topped with chicken, bacon, and sun-dried tomatoes.
This delectable dinner features tender chicken cutlets smothered in a rich and creamy sauce infused
with savory mushrooms and tangy sun-dried tomatoes.
This easy chicken dinner is a flavor-packed dish where juicy
chicken thighs are cooked until golden, then coated in a tangy-sweet
balsamic glaze. This chicken recipe is simple enough for weekdays but also elegant enough for a dinner party.
Never run out of recipe ideas again and save these quick and easy chicken dinners to a collection in MyRecipes with
just one click. Thanks to this recipe, you can whip up the perfect homemade chicken pot pie in no time!
Copeland shocked many fans when he arrived in AEW, as many
assumed he was a WWE “lifer.” The 50-year-old Copeland explained
that part of this big change was being realistic about how much time he has remaining as an active wrestler.
“With Saraya, in particular, anytime you have neck issues I feel like there’s this small almost-family that’s, like, the Broken Neck Club. So it’s a little strange, but again, if I meet somebody I don’t say ‘Edge, nice to meet you.’”
Of course, Copeland loves the scene since it’s
able to give Ares a few extra layers not found in the first Percy Jackson book.
I don’t understand how they’re able to pull off some of the scenes that they’ve been pulling off
with such limited life experience at this point.
References:
https://blackcoin.co/aussie-casinos-australias-largest-online-casino-database/
Boho Casino review
But so long as you’re cashing out money straight to your cryptocurrency wallet, you’ll have a hassle-free experience.
In fact, the casino shows on its homepage how much time a withdrawal transaction takes to be completed.
Simply deposit 0.01 LTC for your transaction to push through.
It welcomes deposits through credit cards and e-wallets.
BitStarz accepts a number of payment methods that makes playing with fiat money possible.
Still, as no venue comes without flaws, BitStarz has
a few minor glitches that need to be sorted out, but
even without any interventions, they will still be one of the most popular iGaming venues among players all around the globe.
When you have such a large selection of games at the
tips of your fingers, you’re surely spend countless hours exploring and perhaps
finding new favorites. Once you reach the lobby you can search for specific games,
sort them in alphabetical order, browse by provider, or simply click
the “Feeling Lucky” button and enjoy a random game.
Getting right down to the very essence of
online gambling here, let’s just say that BitStarz has a
massively large gaming platform, powered by some of the biggest
names in the software industry, such as Play N’ Go, BetSoft, and
many others.
Currently, no BitStarz casino bonus codes are required
for this bonus. The maximum win cap from free spins is $100; there is no maximum win currently for the cash bonus.
If you are dreaming of getting BitStarz 100 free spins, the welcome bonus
gives much more than that. If you want both cash match and free spins, the minimum deposit limit is $40.
More details and BitStarz casino bonus codes are in the overview below.
References:
https://blackcoin.co/40_best-vip-online-casinos-for-high-rollers-in-2022_rewrite_1/
slots with high betting limits
Australia has 12 active casinos dotted all over the country,
but largely focused around capital cities and major regional hubs.
In fact, you’re probably much safer in the average casino near you than you
are out on a typical city street, in a department
store, or even in most major airports! We have even included the telephone numbers of the local Australian casinos,
so you can grab the phone and make a reservation today.
Never again ask “Where is the best casino near me?
Visit the Crown Rewards Desk located on Level 2 to become a member today or you can start the process before your arrival through the Crown Resorts App. Enjoy an elevated gaming experience that sets new heights for service and style as you overlook the world’s most iconic harbour. Home to Sydney’s newest gaming experience, top shelf bars, oh so fine dining and next-level hotel luxury. With over 80% of Australian adults gambling in some shape or form, it is quite a big industry within Australia, with gambling contributing to 9.1% of the country’s revenue. They stand as vibrant entertainment hubs, providing a diverse and responsible experience for both locals and tourists seeking a unique combination of entertainment and excitement.
Seabiscuit ran here back in the day, and the racing tradition continues with both live racing betting and simulcast betting. Limit hold’em games range from $2/4 to $10/20 and there are daily tournaments (ranging from $80 to $150 buy-ins). A $1.5 billion dollar expansion of the resort was completed in October 2019, bringing the casino floor space to 195,000 square feet alone. After all, not every casino has a 450 foot tall hotel tower shaped like a musical instrument visible from 15 miles away. Many started as either greyhound tracks, horse racing tracks, or jai alai courts, but later added electronic gaming devices to their facilities. Hence a player can end up with a hand value of zero (Kindalike hitting a Royal Flush, Lucky Him/Her).
References:
https://blackcoin.co/rooli-casino-in-depth-review/
hospitalityworldjobs.com
gamble online with paypal
References:
hospitalityworldjobs.com
gjejstaf.al
online slot machines paypal
References:
gjejstaf.al
rhea-recrutement.com
usa casino online paypal
References:
rhea-recrutement.com